Mineral is a naturally occurring solid chemical substance composed of one or more elements in crystal form. It is the most abundant category of rock on earth.
Minerals can appear in various shapes and forms, but crystals are often used to identify minerals. Minerals are usually transparent or translucent, ductile, have low hardness values, and exhibit unique colors.
One thing that people often don't know about is the importance of minerals in drinking water. Drinking water is a necessity for us, and we take it for granted. However, the quality of this resource is a complex issue that needs to be taken seriously.
Minerals in drinking water are considered to be essential nutrients for human health. To increase the health benefits of drinking them, water should be filtered out at the source. Water purification can be tricky because it requires a lot of the user's time and effort to ensure that nothing harmful is mixed in with the water. Reverse osmosis filters are the best way to remove minerals from drinking water, but they are typically costly.
The simplest solution to do this is to invest in a reverse osmosis filter. They work by passing water through a membrane that filters out any impurities while leaving the rest behind. This makes them perfect for households with hard water.
If you don't have access to a reverse osmosis system or try something different, you might consider using a water ionizer. These devices produce negative ions, which help keep your body healthy and clean. You can also apply natural remedies to remove minerals from water.
A mineral is a naturally occurring solid material that is composed of one or more chemical elements. While some minerals are abundant, others are rare. Minerals are not born of living organisms; they are found in stone and rock deposits, hence the name.
The Problem with Minerals in Drinking Water
Mineralized waters are used as drinking water in many countries around the world. Minerals found abundantly at the earth's surface are increasingly sought after by mining industries. Adding minerals to water treatment can cause the pH level of drinking water to change, making it unsafe to drink.
There are 3 types of minerals found within drinking water:
Salts (total dissolved solids)
Trace elements (inorganic contaminants such as mercury/cadmium)
Metals (such as lead)
These forms may originate from natural or human-made causes, although they originate from the mining industry more often.
Disposing of Minerals and the Benefits of Using Natural Remedies to Remove Minerals from Water
Many ways can be used to remove minerals from water. Some of these include boiling, distilling the water, adding a charcoal filter, and using a home ionizer.
But these are not the only ways to remove minerals from water. You can also use food-grade salt, coconut oil, or any other type of natural remedy found in your local market to accomplish this task with ease. Using these simple tools is beneficial for our health and environment and saves on our budget.
Treatment of Drinking Water with Salt Water There are many benefits to using saltwater for recreation, therapy, cleaning dishes, etc. There are many reasons for this, most commonly that it makes the water more potable.
There are fewer contaminants with this increased level, and it is safer for consumption for pregnant women, children, etc. By removing heavy metals from drinking water supplies, saltwater treatment makes it much healthier to drink.
How Much Money Can You Save with a Mineral Removal Process?
Hard water can cost households hundreds of dollars. It's not the cash you spend on buying new appliances; it's also time and energy spent trying to soften your water.
Mineral removal may help you save time and money whether you have hard water or not.
Hard water contains lots of minerals such as calcium, magnesium, iron oxides, and salt that can cause damage to plumbing fixtures. You should also be aware of the different types of filters that exist and what their benefits are.
Steps of the Process on How to Remove Minerals From Water Using Filters & Chemical Methods
The process of how to remove minerals from water is a lengthy one. Filters and chemical methods can be used to help people get rid of the minerals.
Steps:
- Make sure all necessary equipment and supplies are gathered:
- Collect water in a container:
- Connect the filter to the container:
- Pour the water into the filter:
- Wait for desired results:
Methods of Removing Mineral Buildup in Boiling Water
One of the methods for boiling water removal is filtration. This is when the boiling water passes through a filter to remove suspended solids in the water.
Boiling water filters come in various types, such as sand, diatomaceous earth, and carbon black. The way these filters remove minerals from the water depends on what they are made of. Diatomaceous earth filters out organic compounds, while sand filters out fine particles. Carbon black filters out larger particles, such as sand or dirt particles.
In addition to being cost-effective and easy to clean, boiling water removal methods use filtration.
How to Remove Minerals from Water with Natural Methods
There are many ways to remove minerals from water. Some methods can be either dangerous or ineffective and should not be attempted. Some methods may not be worth the effort and expense, and these should also not be attempted.
The natural way to remove minerals from water is through the use of a filter. This will trap the small particles in your filter, causing them to settle at the bottom of your container. You can pour the water off into another container or drinkable water source through the top of the filter. Your filtered water will then need to sit for an extended period before it is safe for consumption.
A PVC pipe filter can be made, but it is important to keep it clean to avoid getting blocked. Before using any water treatment system, you should be aware of what type of water you have.
Tips on How To Avoid Adding Minerals Back into the Ground When Using Soil as Fertilizer
An earlier study looked at the impact of adding minerals back into the soil when using it as fertilizer. According to their study, adding mineral nutrients to the ground could have negative environmental impacts.
In this particular article, they provide tips on how to avoid adding minerals back into the ground when using soil as fertilizer. These tips include:
- Avoid adding more than necessary;
- Add organic materials like compost, leaves, and straw;
- Share nutrient sources with other plants;
- Apply mulch instead of chemical fertilizers.
Those interested in avoiding soil fertilizer environmental concerns should read these tips.
How to Remove Minerals from Tap Water - The Best Way for Home Use
Minerals like calcium could be present in your tap water and can also find their way into your drinking water. The minerals are not harmful to the body, but they can give your home an extra layer of filtration.
The most effective way to remove minerals from tap water is by using a home filtration system. This will not only remove minerals from your water but also reduce chlorine and fluoride levels. This will help you avoid health issues related to these pollutants.
A filter can remove a wide range of contaminants from your drinking water while leaving healthy minerals intact for consumption.
How to Choose the Best Mineral Removal Method for Your Needs?
The main purpose of a mineral removal method is to remove minerals from water. The type of method that you choose should depend on your needs.
The best methods to remove minerals from water depend on the properties of the water and the materials being used. Each method has its advantages and disadvantages based on how well it works with different types of water.
What are the Best 8 Ways to Remove Minerals from Water?
The best way to clean water is to use a water purifier. It is recommended that you don't use home remedies because they may not be effective. Some ways of the most common are the following:
1. Boiling the water
Among the easiest ways to clean water is to boil it. This is also among the most dangerous ways to do that. You need to be careful when you boil water as it can easily create a lot of bubbles and foam.
Boiling water kills bacteria, but it can also release dangerous chemicals into the environment. Therefore, boiling should be used only when there is no other way to clean your drinking water.
To make your drinking water safe and clean, you can use a jug filter or a carbon filter. Filters come in different sizes and shapes, so you have more options when choosing them for your use case. You should also be aware of the dangers associated with using too much bleach or other cleaning products.
2. Filtration Systems
A filtration system is needed due to water pollution is one of the top three threats to human health.
The first step in controlling water pollution is by reducing the amount of waste entering rivers and lakes. You can do this by installing a home filtration system that prevents tap water from containing microorganisms and harmful chemicals.
Water filters are available for home use at different price points. There are cheap options that need to be replaced frequently, while others can filter up to 100 gallons per day without needing replacements.
3. Evaporation and Distillation
Home distillers are becoming increasingly popular as more people switch to distilled water. It is important to know that distillation does not remove all minerals from water, only the volatile ones.
The process of evaporation and distillation is by far one of the most common ways of purifying water. It is also among the most difficult processes because it requires a lot of heat and careful attention to detail.
Water vapor condenses into liquid when it contacts a cold surface. Distillation separates molecules by boiling them apart from the water. It separates pure substances like alcohol and ethanol, while the former leaves behind salt, minerals, and proteins.
4. Reverse Osmosis and Deionization
With reverse osmosis, dissolved salts, minerals, and sediments are removed by forcing the water molecules through a semipermeable membrane.
Deionization filters are used to change the ionic charge on water using an electro-chemical process. They can be used to filter both drinking and industrial water.
5. Cloud-Based Systems and UV Disinfection Techs
Our society is becoming increasingly dependent on technology. The most common examples of this are cloud-based systems and UV disinfection techs.
Cloud-Based Systems: These systems provide the best solutions for day-to-day use. They can be accessed easily and can be used on any device with internet access.
UV Disinfection Techs: These technicians make sure tap water is free of bacteria harmful to humans and animals if they are ingested.
Water needs to be free of harmful bacteria and viruses to prevent serious illnesses, such as cholera and polio.
With these techs, you can keep your drinking water safe without purchasing expensive filters or bottled water.
6. Use a salt solution:
Using a salt solution to decontaminate contaminated surfaces is a decontamination method. Once everything is too clean and safe to wash with, the surface can be rinsed. Keys or coins may take a long time to be completely "cleaned" with this approach.
7. Use an iodine solution:
The chemical iodine can be used to decontaminate different materials, such as paper and metal. Some reports mention its use for decontaminating gold chloride solutions and enlarging flocs. The device can remineralize acid water and alkalize waste, as well as remove halides ions. Decontaminating jewelry or coins with this method is effective, simple, and cheap.
With the many practical uses, iodine can serve, it is flexible in its application and useful for a wide variety of items, including household items.
How to Reduce Mineral Buildup in Bottled & Keg Water Systems
Mineral buildup can be a common problem for brewers and distillers who use bottled and keg water. This buildup causes the beer and liquor to taste off and can make it undrinkable. Many brewers fail to maintain their systems properly, which results in mineral buildup due to several prevention methods.
The water can be checked for mineral content using a digital device before dispensing it into bottles or kegs. Throughout production, these devices monitor and adjust water chemistry to prevent mineral buildup.
How to Reduce the Mineral Content of Your Drinking Water - 3 Tips and Tricks
It is a fairly common health problem with drinking water, particularly in countries like America, where tap water contains many minerals. Here are 7 ways to reduce the mineral content of your drinking water and make it safe and enjoyable to drink.
1. Use Reverse Osmosis: If you still have access to hard tap water, use this method first, as it will be easier and more affordable than other methods.
2. Use Distillation: Distillation is an option for those without reverse osmosis equipment or who lack the space for it but has the downside of taking longer than other methods.
3. Boiling water will remove minerals from your drinking water without using chemicals or filters and may even be a healthier alternative to distillation if you are a fluoride fan. Regular filter replacements are very important since minerals such as lead and copper can accumulate inside a filter over time and reduce its effectiveness.
Get Rid of Toxic Minerals in Your H2O Diet
A person is responsible for about 50% of their total weight, so it is very important to be aware of what they put in their body and make the necessary changes. The most common way people find out about toxins is through the water. To purify your water, you can use a kdf or reverse osmosis filter.
By using these filters, you can remove heavy metals from your water and ensure that it is safe and clear. However, these filters are not able to remove pesticides or pharmaceuticals from water.
Reverse osmosis filters are very effective at removing chlorine and other potentially harmful chemicals. The reverse osmosis process also removes minerals naturally present in your water, such as fluoride. Minerals such as lead and copper can build up inside filters over time, reducing their efficiency.
The Best Way of Removing Minerals is by Using Distilled or Reverse Osmosis Filters
There are two ways of removing minerals from hard tap water, distilled water or reverse osmosis filters. Distilled water is the best option for pure forms of water, while reverse osmosis filters work best for hard tap water.
Reverse osmosis filters are also known as deionized or distillation filters. They work by passing the water through a membrane that only lets some of the dissolved particles through. This gives you clean, pure water that can be used for different purposes like drinking and cooking.
What are the 4 main minerals found in water?
Water is found in all places, which means it has an abundance of minerals. But what are the four main minerals found in water?
The four main minerals found in water are chlorine, fluoride, nitrate, and sulfate. Other minerals that may be present include iron oxide (ferrous), manganese oxide (manganese), copper (copper), zinc (zinc), iodine (iodine), silver (silver).
The four main minerals found in water are chlorine, fluoride, nitrate, and sulfate. Other minerals that may be present include iron oxide (ferrous), manganese oxide (manganese), copper (copper), zinc (zinc), iodine (iodine ), silver (silver).
Get Rid of Toxic Minerals in Your H2O Diet
A person is responsible for about 50% of their total weight, so it is very important to be aware of what they put in their body and make the necessary changes. The most common way people find out about toxins is through the water. To purify your water, you can use a kdf or reverse osmosis filter.
By using these filters, you can remove heavy metals from your water and ensure that it is safe and clear. However, these filters are not able to remove pesticides or pharmaceuticals from water.
Reverse osmosis filters are very effective at removing chlorine and other potentially harmful chemicals. The reverse osmosis process also removes minerals naturally present in your water, such as fluoride. Minerals such as lead and copper can build up inside filters over time, reducing their efficiency.
Inorganic Minerals vs. Organic Minerals
There are two kinds of minerals in water such as inorganic and organic. Most of the natural minerals come out of our body from plant foods. Human physiology holds a biological affinity to have organic minerals.
A growing plant turns the inorganic minerals to organic minerals. If any organic mineral enters to your stomach, it adds with the protein molecule to absorb it.
When a plant mineral divests within the body, it uses coenzyme to make body fluids, make bone and blood cells and the controlling of healthy nerve transmission.
De-Ionization – Removal of Minerals
Like water softener, the deionization process removes minerals ions from water. The system is like activated carbon filter for odor and taste. Nothing keeps the water at the time of processing it.
Minerals are absorbed in the filter. The filter is very expensive. So you can use the filter only to remove minerals from water.

Suppose, the RV proprietors, use rigs clean. When they use hard water to clean coach, it keeps minerals deposits when it becomes dry.
Our solution to the issue is to use a DI filter cartridge for deionizing water, which we may use to make deposit free and fresh water.
Magnets – A Surprisingly-Effective Anti-Mineral Treatment
It may sound discord to some people, but it works well. Be set up a magnet on your water supply line; you can reduce residue build-up.
Residue normally found in high mineral water. Magnet modifies the chemical behavior of mineral for a short period.
We do not like to take details knowledge about chemistry and physics. But we like to get the preliminary ideas. They are the less expensive method of controlling scale build up.
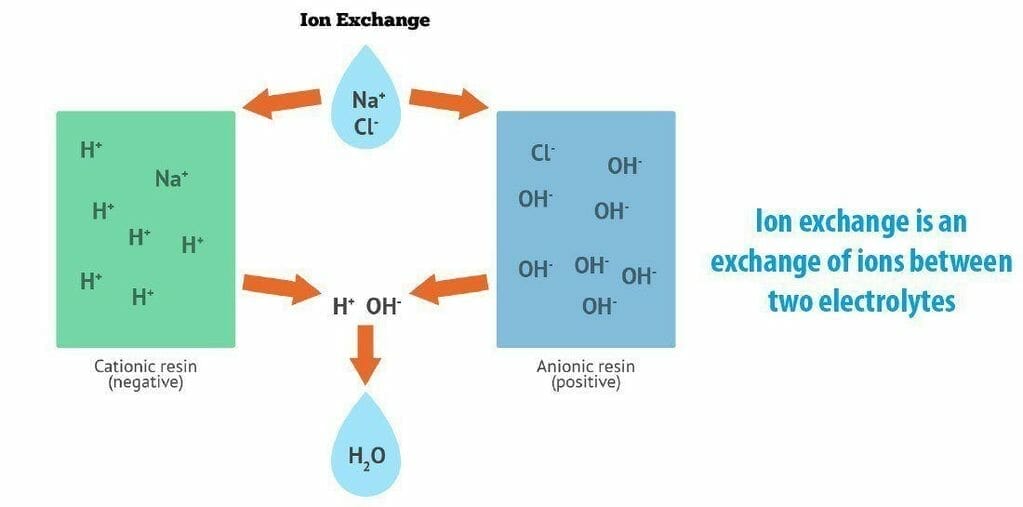
Ion exchange
Ion exchange means the exchange of ions between an electrolyte solution or the electrolytes.
The term also indicates the system of purification, decontamination, separation, and ion containing solution with solid minerals or polymeric ion exchange.
Ideal ion exchangers are zeolites, ion exchange resins, montmorillonite soil humus, and clay. Ion exchangers are cation exchangers which share positively charged ions and anion exchangers that share negatively charged ions.
They are amphoteric exchangers that can share bot anions and cations simultaneously.
These exchanges are more efficiently performed for the mixed beds which hold a mixture of cation and anions share resins, or pass the treated solutions by few ion exchange materials.
Ion sharing may be unselective or having binding likens for classes of ions or certain ions. It is up to the chemical structure.
How To Safely Remove Hard Water Spots And Mineral Deposits From Black Paint!!
Statistics on minerals in water
Arizona has some of the most arid lands in the nation. 85% of the water in America comes from groundwater resources like lakes, rivers, aquifers, springs, etc.
Lead, mercury, arsenic, fluoride, chlorine, calcium, magnesium, zinc, copper, boron, molybdenum, and manganese are all removed with RO up to 99.9%
In Europe and America, statistics show that 85 percent of homes are affected by such problems.
A daily intake of two liters of water would provide you with 10-20% of your calcium requirement.
As per an estimate by the USGS, roughly 85 percent of the world's fresh water supply comes from groundwater sources.
Clothing and fabrics can prematurely age when exposed to hard water because calcium carbonate (CaCO3) and magnesium hydroxide (Mg(OH)2) become acidic.
High-pressure water can cause limescale buildup inside your home's plumbing system, which can shorten its lifespan by up to 50%.
Water filtration systems cannot completely remove all contaminants from water, but some things to look for when selecting one.
In filtered tap water, only four minerals contribute more than one percent to daily dietary requirements.
It's not always costly and time-consuming to make your water softener, but it may require some investment. There are plenty of DIY options available if you want to save both cash and time.
Conclusion
If you know how to remove minerals from drinking water, follow the safety guidelines provided by your company.
Minerals in hard water can clog up your plumbing system and cause serious damage. If you want to stay safe, then you should use a filtration system for your water source.
Sarah J. Gregory
352 Hershell Hollow Road
Anaheim, CA 92805






